




1 / 5











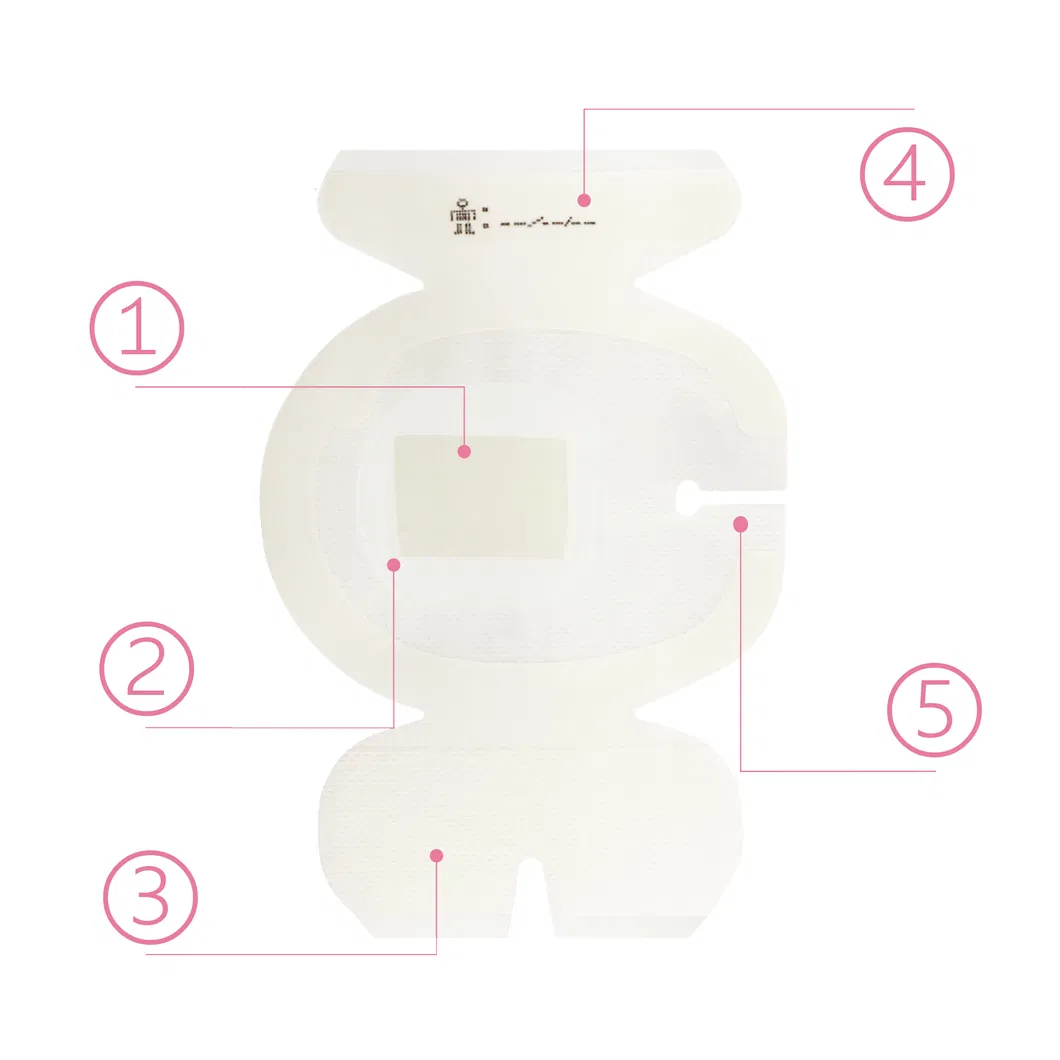
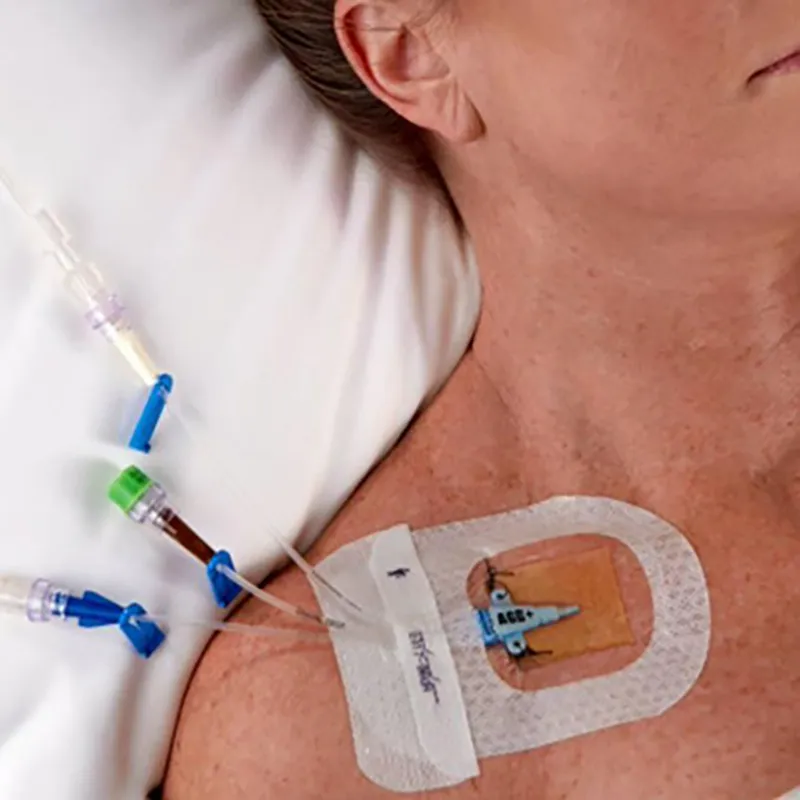
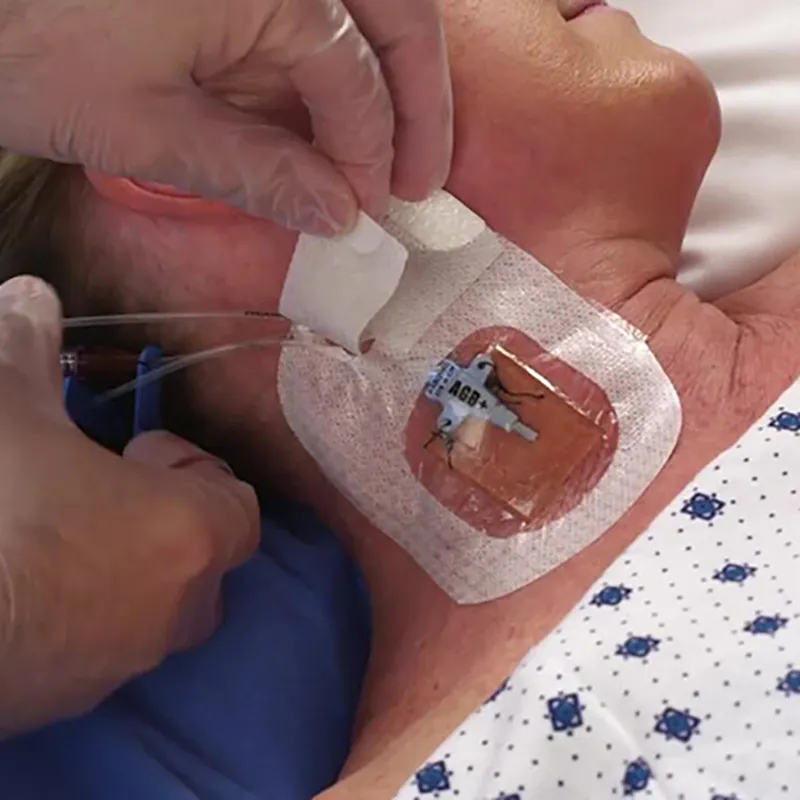
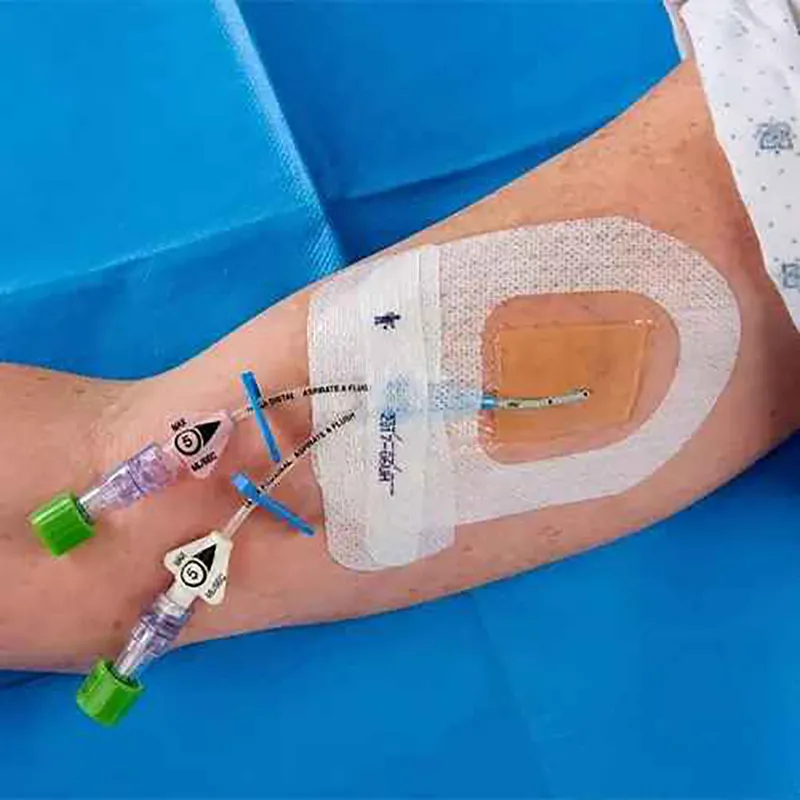
CHG I.V. dressing is composed of a transparent dressing and a gel pad. The gel pad contains 2% chlorhexidine gluconate (CHG), which is an antibacterial agent.
The dressing is used to fix and cover intravenous catheters, other intravascular catheters and percutaneous devices. It can also be used to reduce the number of skin colonies and inhibit the re-growth of microorganisms.




1. Do not use on ulcerated wounds or unhealthy skin.
2. Disposable and external use only; avoid contact with ears, eyes, mouth, or mucous membranes.
3. Patients allergic to chlorhexidine gluconate must not use this product.
4. In case of irritation or systemic allergic reaction, stop use immediately and consult a doctor.


